Sulphur Oxides Emissions
Key Messages
- Canada earns a “B” grade and ranks 16th out of 17 countries.
- Canada’s per capita sulphur oxides emissions are nearly 17 times that of the best performer, Switzerland.
- Canada’s sulphur oxides emissions decreased between 1990 and 2005.
Putting sulphur oxides emissions in context
Air quality is affected by sulphur oxides emitted from smelters, electricity generators, iron and steel mills, petroleum refineries, and pulp and paper mills. Smaller sources include residential, commercial, and industrial space heating. Sulphur oxides contribute to poor air quality in cities and outlying areas, harming human health and quality of life. They can also hurt aquatic ecosystems, buildings, crops, and forests.
How do Canada’s sulphur oxides emissions compare to those of other countries?
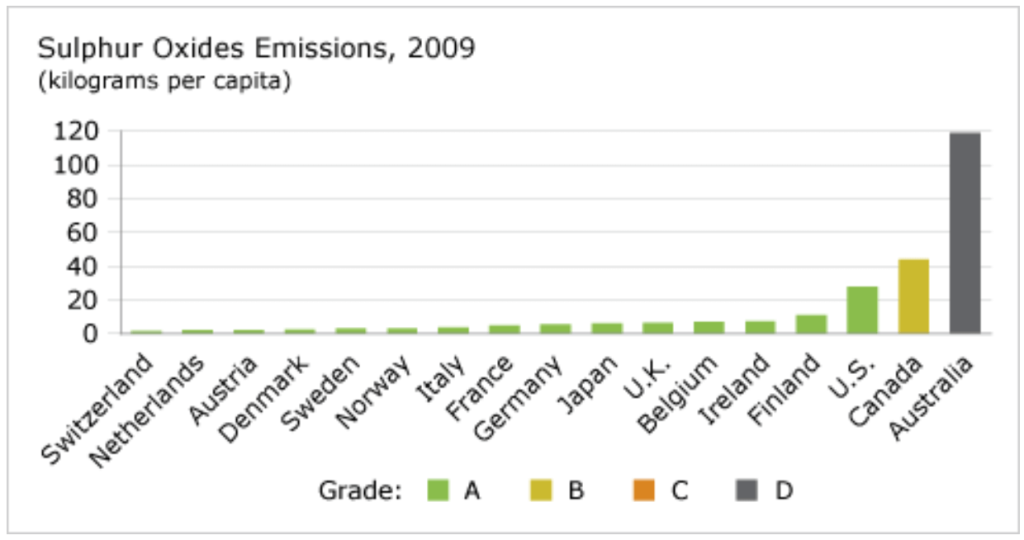
Canada earns a “B” grade for sulphur oxides emissions and ranks 16th out of 17 peer countries. Only Australia receives a lower ranking.
Switzerland is the best performer, with nitrogen oxides emissions of 1.7 kilograms per capita in 2009. Canada’s per capita emissions were 26 times greater than Switzerland’s, amounting to 43.9 kilograms. Australia, the worst performer, had per capita emissions nearly three times those of Canada.
Is Canada’s performance improving?
Between 1990 and 2009, Canada decreased its per capita sulphur oxides emissions by 34 per cent. While any reduction is good, Canada’s progress was weaker than the progress made by 14 of the 17 peer countries. For example, Denmark and Germany each reduced their per capita emissions by 92 per cent, while the U.K. reduced its per capita emissions by 90 per cent. Only one country—Australia—increased its sulphur oxides emissions between 1990 and 2009.
Increases in Australia’s emissions can be attributed to its thermal electricity generation mix, which is dominated by fossil fuels, in particular coal. Demand for electricity in Australia grew at a consistent rate. Large industrial emitters such as the mining and metal industries also contributed to the increase in emissions.
Has Canada’s report card on sulphur oxides emissions improved?
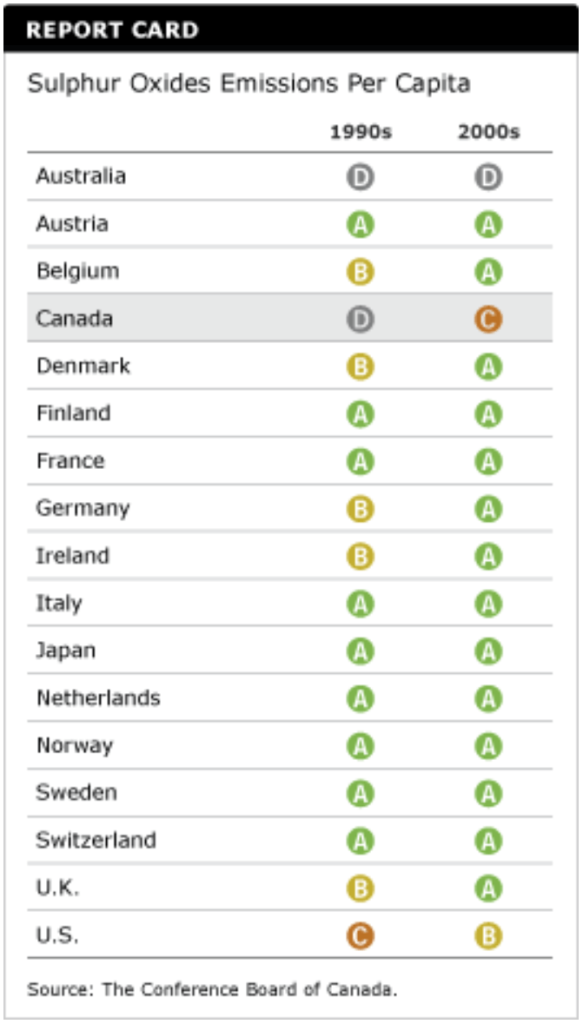
Canada’s grade improved from a “D” in the 1990s to a “C” in the 2000s. This was due to declining per capita emissions in Canada coupled with increasing emissions in Australia. Australia’s increase meant that it took less effort for Canada to get a “C.”
Belgium, Germany, Ireland, and the U.K. improved from “B”s to “A”s. The U.S. improved to a “B” grade.
Where should Canada look for a role model?
The European peer countries have long outperformed both Canada and the U.S. on reductions in sulphur oxides emissions. Like Canada, the U.S. has reduced its per capita emissions by less than most of it peers.
The Europeans have shown that substantial reductions in sulphur oxides are possible over a relatively short time period. In 1990, for example, Germany and the U.K.’s annual sulphur emissions exceeded Canada’s by a significant amount. By 2009, however, both countries had reduced their emissions to levels below that of Canada.
How has Germany managed to reduce emissions so dramatically?
Germany’s reduction in SO2 emissions has been due to replacing old combustion facilities with new ones that have the best available technology. Germany has also regulated desulphurization of flue gases in large combustion plants in the eastern part of the country and switched from solid to gaseous and liquid fuels. Since 2001, the national government has gone further, encouraging the consumption of low-sulphur fuel for vehicles by offering a tax discount.1
The result? Germany decreased its total annual SO2 emissions by nearly 5 million tonnes within 20 years. In 2009, its emissions were 1 million tonnes lower than those in Canada.

What has been done to reduce sulphur dioxide emissions in Canada?
Canada has ratified the United Nations Convention on Long-range Transboundary Air Pollution (LRTAP Convention), which has three international protocols to reduce SO2 emissions. The first, the 1985 Sulphur Protocol, adopted a flat-rate target to reduce national annual sulphur emissions by at least 30 per cent between 1980 and 1993. The next two, the 1994 Sulphur Protocol and the 1999 Gothenburg Protocol to Abate Acidification, Eutrophication and Ground-level Ozone, are based on effects.2 They aim to reduce sulphur emissions where environmental effects are most severe.
The 1985 Sulphur Protocol called for Canada to cap national SO2 emissions permanently at 3.2 million tonnes by 1993. Canada met this cap in 1992, with national emissions of 3.1 million tonnes. The 1994 Sulphur Protocol allows emission reductions to be geographically targeted to achieve maximum environmental benefit. This approach is important for large, sparsely populated countries like Canada.
Canada has met all its current protocol commitments, including capping regional SO2 emissions in the sulphur oxide management area (which includes parts of Quebec and Ontario, and all of New Brunswick, Nova Scotia, and P.E.I.) at 1.75 million tonnes by 2000.3
Have there been “Made in Canada” solutions?
In 1998, Canada’s energy and environment ministers signed The Canada-Wide Acid Rain Strategy for Post-2000 to address the remaining acid rain problem in eastern Canada and to ensure Canada meets its international commitments to reduce emissions that cause acid rain. The strategy outlines actions to achieve critical loads (levels of acid rain that do not cause harm), establish new SO2 emission reduction targets in eastern Canada, and pursue further reduction commitments from the United States.4
In 2000 and 2001, Ontario, Quebec, New Brunswick, and Nova Scotia established new provincial targets for SO2 reductions under the strategy.5 In the future, targets and timelines will be developed for Canada’s sulphur oxide management area as part of the UN Protocol to Abate Acidification, Eutrophication and Ground-level Ozone. To ratify the protocol, Canada must specify emission ceilings for SO2, nitrogen oxides, and volatile organic compounds.
How do sulphur oxides affect the quality of life of Canadians?
Sulphur oxides can directly harm human and animal respiratory systems, triggering respiratory illness and worsening existing respiratory and cardiovascular diseases. When dissolved by water vapour to form acids, it can further harm respiratory systems.
Combined with nitrogen oxides, these gases react with other substances to form acid precipitation, in the form of rain, fog, or snow. Acid rain damages vegetation, buildings, and materials, and it contributes to acidification of aquatic and terrestrial ecosystems.
When transformed into sulphate particles and combined with other particles in the atmosphere, SO2 can contribute to the secondary formation of particulate matter (PM2.5). PM2.5 harms human health and the environment, and contributes to visibility impairment and regional haze.
Footnotes
1 V. Vestreng et. al., “Twenty-five years of continuous sulphur dioxide emission reduction in Europe,” Atmos. Chem. Phys. Discuss, 7 (2007), 5119.
2 United Nations Economic Commission for Europe, Convention on Long-range Transboundary Air Pollution, March 14, 2008 (accessed August 19, 2008).
3 Environment Canada, What’s being done?, December 19, 2002 (accessed August 19, 2008).
4 Canadian Council of Ministers of the Environment, Five-year Review of the Canada-Wide Acid Rain Strategy for Post-2000 (Ottawa: Canadian Council of Ministers of the Environment, December 2005).
5 Canadian Council of Ministers of the Environment, Five-year Review of the Canada-Wide Acid Rain Strategy for Post-2000 (Ottawa: Canadian Council of Ministers of the Environment, December 2005).

